Water is absolutely essential to life from both a biological and physical standpoint- our bodies are mostly water, and the water vapor in our atmosphere helps to keep our planet livably warm. Learn where the earth’s fresh water resides!
Introduction
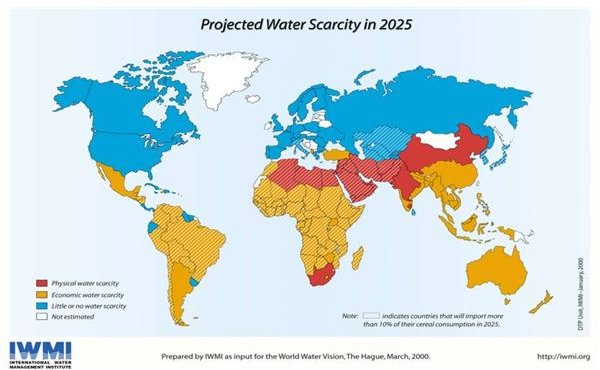
Even though water would seem to be an abundant resource, the truth is that it is far more rare than people in wealthy countries seem to realize, though recent droughts and fights over water rights in the US suggest that such attitudes may be changing. While there are approximately 1.4 billion cubic kilometers of water on earth, most of that is contained in the oceans and undrinkable without significant effort. For water supplies, humans rely on rivers, lakes, and groundwater.
Groundwater
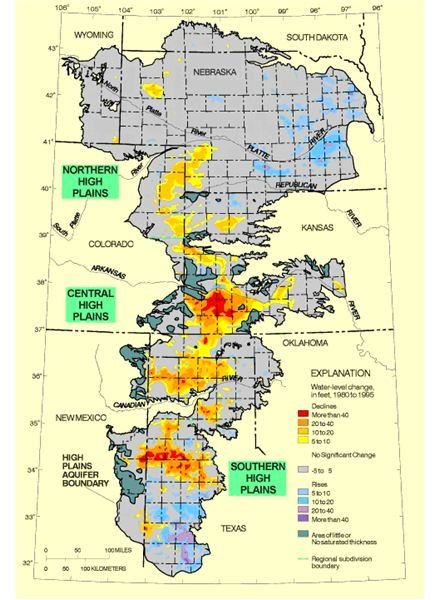
Aquifers, underground “lakes” or concentrations of water, contain the lion’s share of the world’s water supply, perhaps as high as 97 percent of it. Indeed, the Ogallala aquifer that underlies Nebraska, Kansas, Oklahoma, Colorado, New Mexico, Texas, South Dakota, and Wyoming contained an estimated one quadrillion gallons of water in 1980; since then, the water table has dropped by about two feet per year. While water withdrawal via well does not necessarily pose a risk to that water supply (except in terms of depleting the aquifer itself), contaminants can seep into the ground (and do so faster in already saturated ground). Such underground water stores typically recharge through areas of soil and/or rock with high porosity. Wetlands are another important source of “free” water filtration and biodiversity that has been largely eliminated in favor of human settlements, though recent legislation combined with grassroots efforts may slowly reverse this trend. In fact, New York State, in trying to cope with pollution issues in the New York City watershed, found that watershed management is a viable and economically attractive alternative to water treatment plants.
Chemistry & Physics of Water
Water is a critical resource for life here on Earth- water makes up 70 percent of most organisms and makes the chemical reactions that we associate with life a possibility. It provides shelter, food, habitat, and recreation. It is the driver of a significant amount of rock weathering, both physical and chemical. Unlike most compounds, water undergoes a reduction in density when it changes to its solid form; without that, there would be no icebergs, since all ice would be at the bottom of its respective ladder. Its virtue as a “universal solvent” and strong surface tension allows it to be the transportation medium for vitamins, minerals, and other compounds needed in the day-to-day activities of biological organisms. It even serves as a greenhouse gas (without any greenhouse effect, the planet would be cold and uninhabitable)!
This post is part of the series: Water, Water…
Water is absolutely critical to the existence of life on Earth, and, for the needs of a significant fraction of flora and fauna, that water must be in the form of freshwater. Where does it come from, what affects it, what issues surround it, and how does it affect organisms? Find out here!