Even scientists don’t completely understand this topic, but they do know that enzymes speed up chemical reactions in the body. Learn about the functions of these chemicals to better understand their importance.
Enzymes are present in all living systems. Chemically, enzymes are proteins but a special kind of protein. In addition to having all the typical properties of proteins they possess a catalytic activity, that is the ability to accelerate chemical reaction rates tremendously.
There are many different types of enzymes and each one is a catalyst for a specific reaction. Examples of where enzymes are used are in the digestion of food, replication of DNA and protein synthesis.
Studies of enzyme reaction rates have estimated that enzymes accelerate reactions by factors ranging from 100 million to 10 exp 20 (1 followed by 19 zeros) times that of reactions that have not been catalysed.
For example - take a common sugar used by plant cells - sucrose. It has to be broken into its components (fructose and glucose) before it can be used by the cell for functions such as the synthesis of cellular components. It can be broken down by acids or by an enzyme called invertase. In the absence of catalysts the reaction is basically non-existent. In the presence of acids (hydrogen ions- H+) the conversion of sucrose to glucose and fructose proceeds at a relative rate of 1.00 (25oC). When invertase is present (such as in a living cell) the relative conversion rate is of 55,800,000,000. How’s that for a speedy enzyme reaction?
Enzymes are synthesized in vivo by living cells based on information contained in specific genes (one gene one enzyme). This makes enzymes extremely complex protein structures. But it is this complexity that makes enzymes so efficient in doing what they do. In fact a defect (mutation) in the gene will produce a defective enzyme that will not function or it will do so at a much lower capacity.
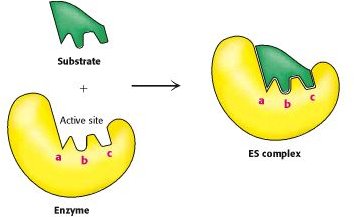
Science has identified several thousand enzymes and there have been studies that have looked at how enzymes work. For a reaction to take place an enzyme binds to a very specific substrate that it is able to recognise. This is referred to as an enzyme-substrate complex. And at this moment the enzyme speeds up the reactive process.
An enzyme has an ‘active’ centre and the substrate (which will be chemically altered in the process) binds to it long enough for a reaction to occur with it and with other substances that are needed to be reacted with.
The Lock and Key Hypothesis of Enzyme Action
So the binding of the substrate to an enzyme has to happen for the enzyme to work. It’s this specificity that makes an enzyme so special. Very early on in enzyme research a useful hypothesis was established to try and understand the molecular cause of this specificity. This was called the Lock and Key Hypothesis. According to it, an enzyme and its substrate are a perfect fit geometrically. The enzyme is the ’lock’ and the substrate is the ‘key.’
Although useful, the Lock and Key Hypothesis is not the complete explanation of how enzymes work and participate in enzyme reactions. Other ideas have been postulated, such as the induced-fit hypothesis in which it is said that upon binding, the substrate binding the enzyme modifies its structure so as to better accommodate the reactive groups, and thus make the reaction easier.
Sources
Whitaker, J. R. 1994. Principles of enzymology. Marcel Dekker, New York.
Knowles, J. R. 1991. Enzyme catalysis: Not different, just Better. Nature350:121-124.
Voet and Voet .2005 Biochemistry. Third edition. John Wiley and Sons, Inc. New York