Antisense technology is still in its infancy, but may prove to be one of the most important breakthroughs in the battle with genetic disorders and diseases.
Antisense technology is a treatment that has been developed to tackle the effects of genetic disorders and infections. Certain genetic sequences within genes can cause diseases, by leading to the insufficient or excessive production of proteins. The concept behind antisense therapy is that it can stop the production of these protein products and thereby cure the disease. Antisense technology uses a synthesized strand of nucleic acid, either DNA or RNA, that can bind with the messenger RNA (mRNA ) that is produced by the gene. The strand causes the mRNA to become inactive, stopping the protein in its tracks. Antisense technology works because in order for mRNA to be translated, it must exist in the form of a single strand.
History of Antisense Technology
The general concept for antisense technology was first created in 1978. It has gone through a variety of technical adjustments over the years. Scientists coined the term “antisense” because the mRNA sequence is known as the “sense” sequence. Since this technology blocks the mRNA it became known as “antisense.”
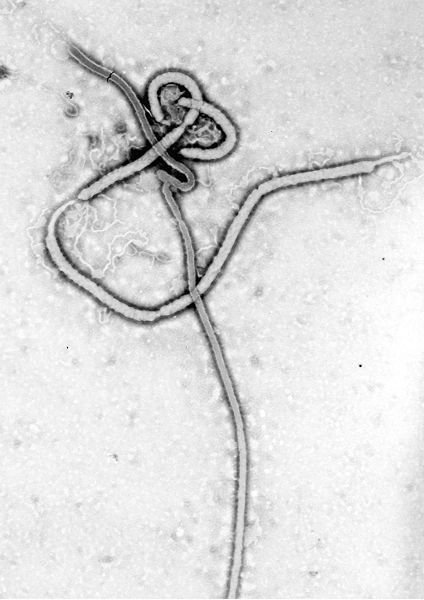
In 2006, the first major breakthrough in this form of therapy occurred. While studying the Ebola virus, the U.S. Army Medical Research Institute for Infectious Diseases found that after using antisense drugs on infected monkeys, 75 percent of the subjects recovered from the disease. This was a paramount discovery because the mortality rate from Ebola is generally at 100 percent.
That same year, German physicians used an experimental antisense drug on patients with a type of brain cancer called gliomas. The results of the research showed that the drug could offer a potential cure for the usually fatal disease.
Working on the success of these and other experiments, many pharmaceutical companies began to experiment with formatting the technology into an approved drug form to help fight genetic disorders and genetic diseases. Isis Pharmaceuticals has become the first company to get an antisense drug approved by the FDA. Vitravene was released in 2008 in an injectable format. The active ingredient in the drug is fomivirsen sodium and its presently being used for patients with inflamed retinas and AIDS.
Genetic Disorders and Antisense Technology
Cells in the human body contain the genetic blueprint for life in their nucleus. With the research conducted by the Human Genome Project , scientists understand that chromosomes within the nucleus contain about 30,000 different genes. Each of these genes acts as a set of instructions to form a specific protein. These proteins are responsible for a number of features in the human body, including genetic disorders and diseases. Traditional drugs can target proteins within the cell and inhibit or enhance their function. Antisense drugs, however, act to prevent the creation of those proteins in the first place.
The compounds in antisense therapeutics need to be designed with the exact nucleotide sequence that will interfere with the target mRNA. Researchers link together short chains of 12 to 30 chemically stabilized nucleotides. These are known as oligonucleotides. These complementary genetic codes enter the cell and attach to the mRNA.. This causes protein production to be inhibited. The ribosomes are unable to read the message the mRNA is attempting to deliver. Some antisense drugs are even able to use an existing enzyme within the cell called RNase H to destroy the mRNA. This enzyme exists naturally to exterminate double-stranded nucleotides, which is how the mRNA appears with the antisense strand.
This means that antisense technology can essentially interact with any one of the human genes, unlike traditional drugs.
structural formula of fomivirsen antisense drug
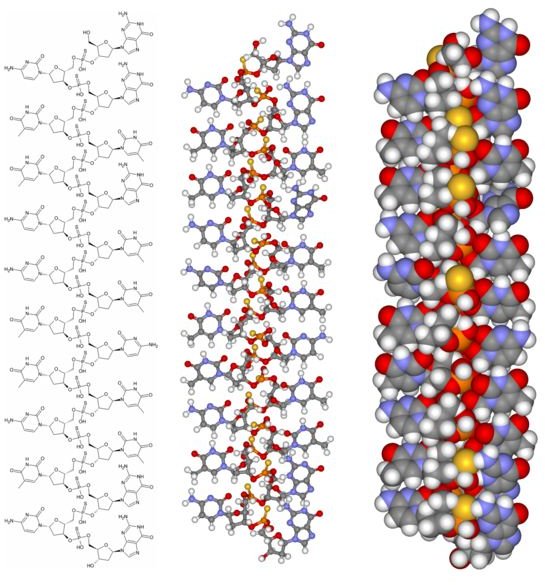
Image Sources
structural formula of fomivirsen antisense drug. (Image credit: Fvasconcellos at Wikimedia Commons, https://en.wikipedia.org/wiki/File:Fomivirsen _composite.png, public domain.)
Ebola virus. (Supplied by the Centers for Disease Control; Public Domain; https://upload.wikimedia.org/wikipedia/commons/f/ff/Ebola _virus_em.jpg)