Biological waste water treatment systems remove organic matter (biochemical oxygen demand) from waste water, utlizing dissolved oxygen by aerobic bacteria. Activated sludge, trickling filter and waste water treatment lagoons are common types of biological waste water treatment systems.
Biochemical Oxygen Demand and the Need for Biological Wastewater Treatment
Organic matter in water will naturally be oxidized to carbon dioxide and water by aerobic bacteria present in the water. The aerobic bacteria (as implied by their name) will utilize dissolved oxygen from the water in carrying out this biological oxidation. If oxygen was extremely soluble in water, then the small amount of dissolved oxygen used to oxidize organic matter in water wouldn’t be a great concern. Unfortunately, however, water becomes saturated with dissolved oxygen at a level only a bit higher than that needed by fish and other aquatic life. See the article, “Biochemical Oxygen Demand (BOD) as a Measure of Organic Water Pollution ,” for more discussion of this topic.
The biochemical oxygen demand (BOD) exerted by organic matter in water, as described briefly above, competes with aquatic life for the precious dissolved oxygen in water, and is the reason that biological waste water treatment systems are a major component of almost any waste water treatment plant. The most commonly used biological waste water treatment systems are activated sludge, the trickling filter, and the waste stabilization lagoon. All three of them treat waste water by bringing aerobic bacteria, dissolved and fine suspended organic matter, and dissolved oxygen together so that biological oxidation of the waste organic matter takes place in the treatment plant rather than in the receiving stream.
Each of the commonly used biological waste water treatment systems will be introduced and discussed briefly in the remainder of this article. See the rest of the series for more details about each process.
The Activated Sludge Process
There are many variations of the first biological waste water treatment system we’ll be considering first, the activated sludge process. It will be discussed here in very general terms and covered in more detail in a later article in this series. The diagram at the left shows a flow diagram for an activated sludge secondary waste water treatment plant. Preliminary treatment typically consists of screening, flow measurement and perhaps grit removal. The primary clarifier removes settleable solids and the primary

effluent then goes to the heart of the activated sludge process, the aeration tank. This is where biological oxidation of the dissolved and fine suspended organic matter in the waste water takes place, due to the juxtaposition of the organic matter in the waste water, dissolved oxygen (maintained by blowing air into the aeration tank through diffusers) and aerobic bacteria (the ‘activated sludge’ being settled out in the secondary clarifier and recycled to the aeration tank). By carrying out the biological oxidation in the waste water treatment plant, the organic matter doesn’t go to the receiving stream and use up dissolved oxygen there.
For more details about the activated sludge process, see the article, “Learn About Activated Sludge Systems in the Treatment of Wastewater .”
The Trickling Filter Process

A trickling filter, biological waste water treatment system will have the preliminary treatment, primary clarifier, and secondary clarifier, as just shown in the activated sludge flow diagram above. The aeration tank, however, would be replaced by a ’trickling filter.’ The trickling filter is a large diameter cylinder, about 6 ft tall, filled with rocks (or perhaps some synthetic medium) and having an underdrain system at the bottom to allow treated

waste water to be drawn off and to allow air to flow in at the bottom and up through the bed of rocks. The trickling filter brings organic containing water, aerobic bacteria, and oxygen containing air into contact, although in a somewhat different manner than the activated sludge aeration tank. The aerobic bacteria are in a slime layer which forms on the rocks; the water is ’trickled’ onto the top of the rock bed, as shown in the two pictures. Air is drawn in through the underdrain system under the bed of rocks and flows up through the bed. Due to bringing the organic matter, aerobic bacteria, and air together, the organic matter is oxidized to carbon dioxide and water.
The Waste Stabilization Lagoon
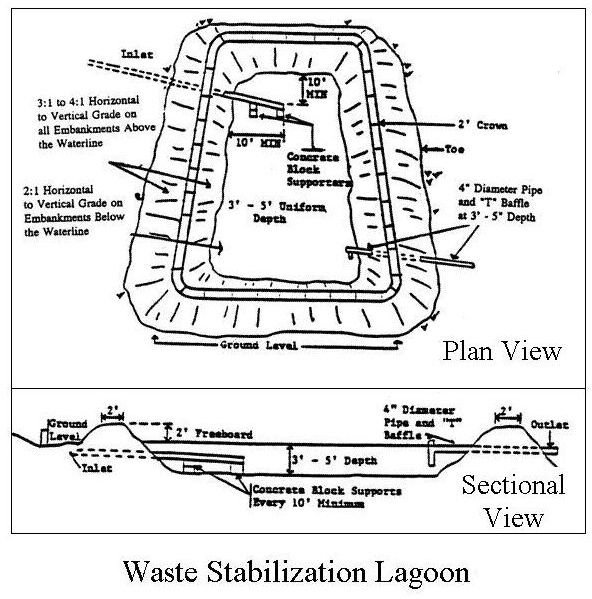
The third biological waste water treatment system is known by many different names, such as: waste stabilization lagoon, oxidation pond, oxidation lagoon, etc. It is most suitable for relatively small flows of waste water,

because it requires more land area per unit of waste water flow, but less operational attention. A waste water lagoon is a constructed pond of appropriate size to retain incoming waste water for about 30 days, with engineered inlet and outlet structures. This allows enough time for the biochemical oxygen demand in the waste water to be oxidized by aerobic bacteria, which thrive in the pond because of the steady supply of ‘food’ being provided. In a properly functioning waste treatment lagoon, algae on the surface of the lagoon produce dissolved oxygen during daylight hours and help to keep the top part of the pond aerobic. The diagram on the left shows a plan and elevation view of a typical waste stabilization lagoon and at the right is a picture showing part of a waste treatment lagoon.
Image Credits
Activated sludge aeration tank: https://nsm1.nsm.iup.edu/tsimmons/Environmental%20Health%20Photolibrary/ENVH _Photolibrary_Wastewater_Images.shtm
Trickling filter picture: https://www.lifesciences.napier.ac.uk/smaefiles/wlinton-broughton/trickling3.jpg
Trickling filter distributor arm: https://www.training.gpa.unep.org/images/jpg/trickling _filter_sm.jpg
Waste stabilization lagoon diagram: https://www.ilga.gov/commission/jcar/admincode/077/07700905ZZ9996arR.html
Waste stabilization lagoon picture: https://www.xr-5.com/case-histories/ch.php?id=25
References
References for Additional Information:
1. Grady, C.P.L., Daigger, G.T., Lim, H.C., Biological Wastewater Treatment, 2nd Edition, New York, NY, Marcel Dekker, Inc., 1999.
2. Metcalf & Eddy, Inc, (revised by Tchobanoglous, G, Burton, F.L., Stensel, H.D., Wastewater Engineering Treatment and Reuse, 4th Edition, New York, NY, 2003.
3. Vesilind, P.A. and Morgan, S.M., Introduction to Environmental Engineering, 2nd Edition, Belmont, CA, Brooks/Cole, 2004.
This post is part of the series: Information on Wastewater Treatment
This series will provide information on wastewater treatment, its processes, and the specific methods used at wastewater treatment plants to remove pollutants from the water.



