Amino acids are the structural components of proteins. But what is the atomic structure of amino acids? This article demystifies and explains the structures and properties of all twenty amino acids.
One of the most important structural and functional components of the human body is protein. Proteins are polymeric molecules made from combinations of 20 different components, known as amino acids , arranged in different sequences. Hence amino acids are the building blocks of all proteins, accounting for almost 75% of our body’s dry weight. This article explores the atomic structure of amino acids.
How Many Amino Acids?
Most textbooks would say that there are 20 amino acids. In reality, there are hundreds of amino acids, but its 20 which are used most commonly in protein synthesis, and form the ‘major amino acids’. The others are either not used by our body or are derivatives or minor variants of any of the 20 major amino acids. Hence these are also known as ‘Minor amino acids’, for example, phosphoserine is a minor amino acid derived from serine, a major amino acid.
Atomic Structure
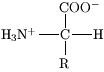
There are many different amino acids in nature, all of them are made of the same fundamental elements - carbon, hydrogen, oxygen and nitrogen. Some amino acids also have sulfur. The basic atomic structure of amino acids consists of a carbon bound to an amine (-NH2) group and a carboxylic acid (-COOH) group, along with a ‘side chain’. This structure is also the reason why the molecules are named ‘amino acids’. The side chain varies amongst all the different amino acids.
The figure shows the general atomic structure of amino acids.
Classification of Amino Acids based on its Atomic Structure
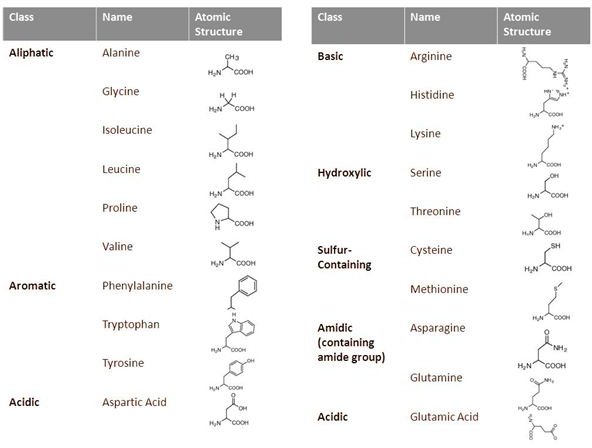
Based on its atomic structure, amino acids are classified as aliphatic, aromatic, acidic, basic, hydroxylic, sulfur-containing and amidic. The Table beside gives the names, class and atomic structure of amino acids (Click to enlarge).
- Aliphatic Amino Acids: These amino acids have an aliphatic side chain in the position R. An aliphatic group is one that is non-polar and hydrophobic and does not include a ring structure. They include Alanine, Glycine , Isoleucine, Leucine, Proline and Valine. The hydrophobicity increases with increasing length of the side chain, with glycine being the lease hydrophobic and isoleucine being the most.
- Aromatic Amino Acids: These amino acids include a six carbon aromatic ring structure. Aromatic amino acids include Tryptophan , Tyrosine and Phenylalanine. They are relatively non-polar and are characterized by their absorbance of UV light. The test for detection of protein, based on measuring UV absorbance at 280nm, makes use of this property of aromatic amino acids, with tryptophan being the amino acid responsible most for this effect.
- Acidic Amino Acids: These amino acids have two free carboxylic acid groups instead of one and are hence acidic and polar in nature. They are easily ionizable at physiological pH. Acidic amino acids are Glutamic acid and Aspartic acid.
- Amide Amino Acids: Each of the acidic amino acids has its corresponding amide form. Hence the two amide amino acids are Glutamine and Asparagine. These amino acids are polar and do not ionize easily.
- Basic Amino Acids:These amino acids have two amine groups instead of one, resulting in a net basic charge. They include Histidine, Lysine and Arginine.
- Hydroxylic Amino Acids: The hydroxylic amine acids include Serine and Threonine. They contain a hydroxyl (-OH) group and are polar and uncharged at physiological pH. The aromatic amino acid tyrosine also contains a free hydroxyl group and may be categorized in this group.
- Sulfur-Containing Amino Acids: The two sulfur containing amino acids are Methionine and Cysteine. These amino acids are critical to formation and maintenance of secondary structure of proteins. Being hydrophobic, cysteine and methionine residues are often found buried deep in the core of the protein.
References
- Lehninger, Principles of Biochemistry, 5th ed.
- Biochemistry by Lubert Stryer, 5th ed.
- Image Credits:
- General Structure of Amino Acids: Ppfk (https://commons.wikimedia.org/wiki/File:Aminoacid _general_structure.png)
- Table of Amino Acid Structures: Prepared by Author