There are different levels and types of methods and applications used to determine protein structure and protein sequence.
Levels of Structure and Their Evaluation
Proteins are found in all living organisms. They thus represent one of the most important classifications of biologically active molecules. Proteins are derived from carbon, hydrogen, nitrogen, oxygen, sulfur and phosphorus atoms. There are a number of methodologies for studying proteins, due to their structural complexity, which is logically divided into four levels.
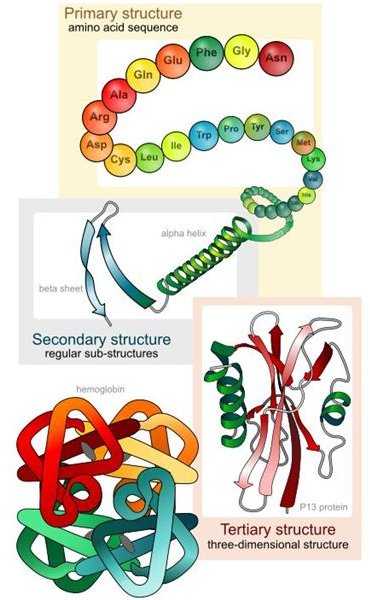
- Primary structure: the basic amino acid sequence of peptide chains
- Secondary structure: the regular alpha helix and beta sheet strands that are defined (there can be different motifs that can be present in a single molecule of a protein).
- Tertiary structure: the three dimensional structures of a protein molecule and the spatial arrangement of secondary structures. Tertiary structure helps describe the polypeptide chain folding.
- Quaternary structure: the complex of many protein molecules and polypeptide chains that are generally called protein subunits and which function as a part of a larger assembly.
All proteins are polymers of amino acids. Such polymers are also called polypeptides and consist of combinations drawn from a pool of twenty unique amino acids referred to as residues. Proteins often alter their structure while performing a biological function. There have been several different methods used to classify the structure of proteins. Each of these helps to classify different data in the protein data bank. There are several different databases that classify proteins.
Approximately 90 percent of protein structure available through the Protein Data Bank has been identified by means of crystallography. This method characterizes protein structure by means of i’s electron density distribution. About 9 percent of protein structure is obtained using Nuclear Magnetic Resonance (NMR) techniques. NMR involves the resonance of the nuclei of atoms in a given protein. This is useful, if difficult, because the nuclei of atoms in proteins each experiences a different environment. If they are different, they are nevertheless similar, so a complex series of evaluations is made to resolve individual resonance peaks.
Since protein sequence techniques are much simpler to use than those defining the higher levels of structure, it is naturally desirable to learn as much employing these methods as possible. This is also true because insight is obtained on how proteins function when they are not in a sequence. Thus there is much return from these simple techniques. Due to the complexities of the other techniques and their application, scientists often resort to computational software modeling of higher protein structure which utilizes the more readily obtained sequence data.
Proteins - Levels of Structure
Credit: Wikimedia Commons by LadyofHats