C-reactive proteins are found in the blood and are present in large numbers during a period of inflammation. This makes the identification of normal C-reactive proteins synonymous with the onset of viral or bacterial infections.
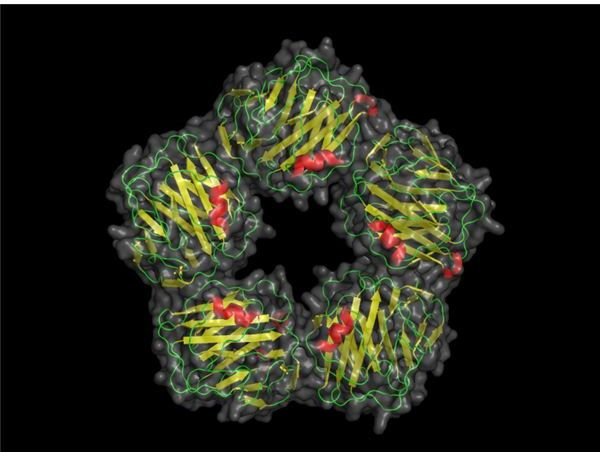
C-reactive proteins or CRPs are found in blood upon the event of inflammation. As a member of the pentraxin family, C-reactive proteins are bound together by calcium into a flattened structure.
CRPs are produced by the liver and fat cells and rise in number when an immune response to inflammation occurs. The proteins bind to phosphocholine, commonly seen as a fatty substance in tissue, and assist in the process of phagocytosis, the consumption of pathogenic cells in the body. Many scientists believe that C-reactive proteins also help general immunity by suppressing early signs of infection.
During an infection, their numbers rise inside the body. The time frame of this occurs within 6 hours and protein number peaks to 50,000 at around 48 hours. Normal C-reactive proteins are associated with serum amyloid A, a similar protein that performs in conjunction with CRPs.
Genetic Base of C-Reactive Proteins
First identified by William Tillett and Thomas Frances in 1930, C-reactive proteins were named after a type of streptococcus pneumoniae extract the team called “Fraction C.” When the disease was administered, the two scientists found that the protein would react to fight against inflammation.
The gene for CRP is located on the first chromosome, 1q23 and is comprised of two exons and one intron. The C-reactive protein is first synthesized as a 206 amino acid polypeptide, then secreted by the hepatocytes as a non-glycosylated monomer with a molar mass of 25106 daltons.
Most scientists see the C-reactive protein structure as a jellyroll structure which forms into a symmetrical unit when it is fully active.
Occasionally, sugars may attach to the CRP. Certain diseases can cause amino acids to abandon its structure. This makes the protein structure vulnerable to glycosylation from galactose, glucose, mannose or sialic acid. This changes the pattern in which the protein is structured slightly and can make the blood cells vulnerable to various parasites. The most common diseases in which this occurs are bone cancer, lupus, leukemia and tuberculosis.
Above: Computer model of C-reactive protein. (Image credit: Skolstoe at Wikimedia Commons, https://en.wikipedia.org/wiki/File:CRP _pretty.png, public domain.)
Use of C-Reactive Proteins in Diagnosis
Outside of liver failure, C-reactive proteins are consistently produced within the body. This makes them prime candidates for charting the effectiveness of treatments to diseases. Because C-reactive proteins act as a marker of inflammation, analysis of blood samples reveal the level of the proteins. If a treatment is progressing well, the volume of the proteins in the blood should decline.
There are various ways to test the blood for the presence of C-reactive proteins. The most common of these is the Enzyme-Linked ImmunoSorbent Assay, or ELISA test. This uses the method of adding an enzyme to the blood that allows physicians to detect the amount of proteins present. This is usually accomplished using a fluorescent compound.
Normal volumes of the protein are less than five milligrams per liter. This figure is amplified during viral or bacterial infection.
Scientists researching cardiovascular disease have identified an association with C-reactive proteins and increased risk of pulmonary failure. Studies indicate that the higher the trans fat levels in a person’s diet, the higher the volume of CRPs. This means that CRPs can be used as an indicator of potential heart problems.
Research has also shown that raised levels of C-reactive proteins can be an indicator of colon cancer (Erlinger TP et al: Journal of the American Medical Association 291 (Feb 4): 585-590). Blood samples reveal that patients suffering from colon cancer have twice the amount of CRPs as regular patients.
References
“Inflammation, Heart Disease and Stroke” American Heart Association (https://www.americanheart.org/presenter.jhtml?identifier=4648)
“C-reactive Proteins” MedLine Plus (https://www.nlm.nih.gov/medlineplus/ency/article/003356.htm)