According to physics, there are four basic forces in the Universe: gravity, electromagnetism, the strong force, and the weak force. To learn more about them, we must look at them from a particle physics perspective.
Gravity and Electromagnetism
Two of the four fundamental forces of nature are readily apparent in every day life and should already be familiar. Briefly:
- Gravity is a force that attracts objects to each other. The strength of gravity increases as mass increases, but it doesn’t have a whole lot of influence on anything until an object gets to planet-sized masses, which actually makes it the weakest of the forces. Beyond those basics, no one is quite sure exactly how it works. The effects of gravity are well-characterized by Einstein’s theory of general relativity.
- Electromagnetism is, basically, electric currents and magnetic fields. The electromagnetic spectrum (visible light, heat, radio waves, UV radiation, X-rays, etc.) is mediated by a particle called a photon.
Both of these have very long ranges that extend well away from their sources. The other two forces have extremely short ranges - much less than the size of the smallest atom. These forces become very important at subatomic levels.
The Strong Force
Most chemical elements have atomic nuclei that contain multiple protons and neutrons - collectively called “nucleons” for being within an atomic nucleus. Protons and neutrons are made of quarks. A proton is two up quarks and a down quark, and a neutron is two down quarks and an up quark.
The strong force holds atomic nuclei together. Protons are positively charged particles, and due to the electric force alone, they should repel each other - but they don’t because the strong force is stronger, hence the name.
Under current understanding in particle physics, the strong force is not so much nucleons clinging to each other, as it is the component quarks of the nucleons clinging to each other via a “color” force. The range of the strong force is just slightly more than the radius of a proton, which gives an upper limit to how many protons can be in a nucleus before electric repulsion takes over. The largest stable atom is an isotope of lead at 208 nucleons (82 protons, 126 neutrons). For more details about the color force, see the first article in this series.
The Weak Force
According to the Standard Model of particle physics, there are twelve known fundamental particles of matter in the Universe. Six of these are types of quarks: up, down, charm, strange, top, and bottom. The other six are called leptons, and include electrons, muons, tau, and three kinds of neutrinos. Among other things, the weak force allows quarks to turn into other types of quarks, and leptons to turn into other leptons. The most well-known consequence of such a change is radioactive decay, which occurs when an atomic nucleus is unstable.
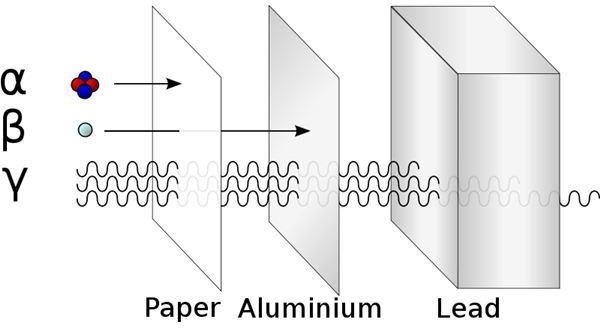
In alpha radioactive decay, the nucleus ejects a cluster of four nucleons - two protons and two neutrons, called an alpha particle (identical to a helium atom). This reduces the total number of nucleons in the nucleus.
In beta radioactive decay, the number of protons in the nucleus changes, which changes the chemical element, but the total number of nucleons stays the same. Either a neutron turns into a proton, or a proton turns into a neutron. More precisely, a down quark becomes an up quark, or an up quark becomes a down quark. When this happens, the neutron emits a W- boson, which then becomes an electron (also called a beta particle) and an electron antineutrino, or the proton emits a W+ boson which becomes a positron (an anti-electron) and an electron neutrino.
References and Photo Credits
Giancoli, Douglas C. Physics: Principles with Applications 3rd ed. Prentice Hall 1991.
Solar System image by NASA.
Lightning image by John R. Southern , used under CC-A-SA 2.0 license.
Quark-gluon plasma image by Brookhaven National Laboratory, used under CC-A-SA 2.0 license.
Radioactive decay image by Stannered , used under CC-A-SA 3.0 license.
This post is part of the series: Particle Physics in Cosmology
The Standard Model of particle physics identifies the elementary particles of matter and provides an explanation for three of the four fundamental forces of nature - electromagnetism and the strong and weak forces, using force mediating gauge bosons and a mass mediating Higgs boson particle.



