Find out about the chemical properties of ibuprofen and how these properties help this drug reduce inflammation.
Ibuprofen is an ubiquitous medication used to relieve pain, reduce inflammation, and lower fevers. It is available both over-the-counter (under many brand names) and, in stronger doses, by prescription. Almost every medicine cabinet contains a bottle of ibuprofen pills. But how does this amazingly useful drug work?
Structure and Properties
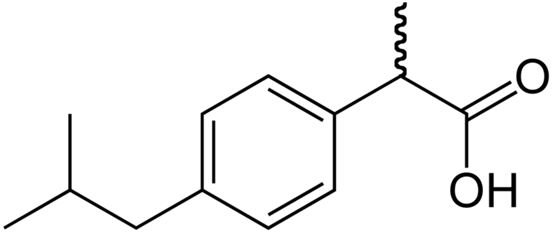
Ibuprofen (C13H18O2) has the systematic name 2-(4-isobutylphenyl)propanoic acid, making it an organic compound in the class of propionic acid derivatives. Its melting point is melting point 74 - 77° C. It is a stable white crystalline powder, slightly soluble in water and very soluble in ethanol. Its formula is also written (CH3)2CHCH2C6H4CH(CH3)COOH, which lays out the chemical structure of ibuprofen.
Ibuprofen is a chiral molecule, meaning that two mirror-image forms (called enantiomers) are possible. In the diagram, the wavy line represents a molecular bond that is bent “upward” for so-called (R)-ibuprofen and “downward” for (S)-ibuprofen. In the human body, only (S)-ibuprofen is active, but an enzyme readily converts (R)-ibuprofen to the active (S)-ibuprofen form. Because of this, drug manufacturers do not bother to separate the enantiomers in their formulations, using a mixture of the two (called a racemic mixture).
How Does Ibuprofen Work?
Ibuprofen is a non-steroidal anti-inflammatory drug (NSAID). It works by inhibiting the activity of an enzyme called cyclooxygenase (abbreviated COX). The COX enzyme converts certain fatty acids to prostaglandins, molecules with important functions in the human body. The prostaglandins at the end of the “chain” of reactions that starts with the COX enzyme cause an increased sensitivity to pain, fever, and vasodilation (increased blood flow or inflammation). By inhibiting the start of this chain of reactions, ibuprofen therefore reduces pain, fever, and inflammation.
NSAIDs are chemically different from the other major class of anti-inflammatory drugs, the corticosteroids. Corticosteroids are powerful drugs that mimic the stress hormone cortisol. Ibuprofen has a different chemical structure and a different mechanism, and its side effects are less severe.
Ibuprofen Chemistry: The Cause of Side Effects
Ibuprofen is notorious for causing stomach pain, nausea and vomiting, and other gastrointestinal symptoms. These side effects are caused by the same mechanism as the beneficial effects. The same prostaglandins that cause pain, fever, and inflammation (which are inhibited by ibuprofen) also decrease stomach acid and increase stomach mucus secretion. Both of these actions tend to calm the stomach. Since ibuprofen inhibits the enzyme that produces these prostaglandins, stomach irritation can result.
References
- Ibuprofen from 3DChem.com.
- Ibuprofen Product Information from ChemicalLand21.com.
- Ibuprofen diagram: Public domain, created by Ben Mills.