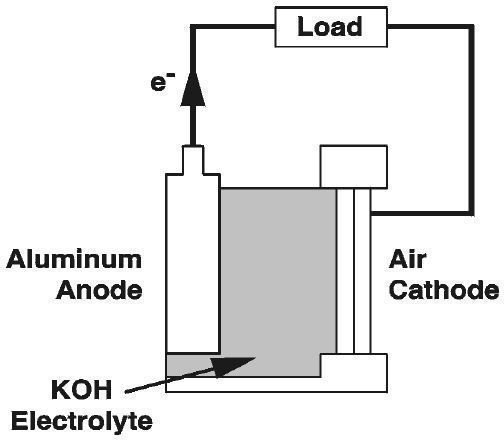
An aluminum-air battery that produces 1 volt and 100 milliamps of electricity can be made from simple household materials: aluminum foil, salt water, and activated charcoal. This can be a great source of power in emergency situations or in places without access to an electrical grid.
When Nikola Tesla first proposed using aluminum air batteries as a way to deliver power to households in America the idea was ridiculed as another of his wild ideas. In reality the power companies that he had helped create with his alternating current (AC) delivery system were the power behind the discrediting of this idea. Again Tesla was a hundred years before his time.
The green movement has embraced homemade versions of the metal-air battery for over twenty years with units like the “energy burrito” offering short term power to eco-minded users in emergency situations. While these homemade devices are easy enough to be made for a middle school science fair, a more advanced model has the potential of delivering much needed electrical power to off gird areas in developing countries or areas with an unreliable electrical infrastructure or lack of solar resources .
Basic Chemistry Behind the Battery
All metal/oxygen batteries work using the same basic chemistry. In the case of the aluminum air battery, the exact chemical reaction is 4Al + 3O2 + 6H2O → 4Al(OH)3. This reaction also nets approximately half a volt of electrical current. Each cell of an air aluminium battery will produce a consistent half volt no matter the size of the individual cell. The size of the cell will however determine how often the aluminum will need to be replaced and the residue cleaned.
The oxidation reaction that takes place in the battery creates aluminum hydroxide gel (a common ingredient in over the counter antacids) that must be cleaned from the surface of the aluminum to allow the chemical reaction to continue.
Potential Capacity
The reason the metal-air batteries are receiving scientific attention is the potential capacity of a single unit. Consider the capacity of a standard nickel based battery with a specific energy of 50 wh/kg(watt hour per kilogram) and a lithium ion battery with a range of specific energy between 85-150 wh/kg. A projected capacity of air-aluminum batteries is 380-455 wh/kg or over three times the capacity of a lithium ion model. Aluminum air battery research has the potential to help develop large scale portable energy sources to be used in remote regions of the Earth.
Real World Uses
Metal-air batteries are already in use- Zinc-air hearing aide batteries are an example. In fact research into metal-air batteries started over 25 years ago. The renewed interest in this time of chemical batteries is due to advances that allow large amounts of aluminum alloy to be produced at very low costs. Who is interested in this technology? The American military, hybrid car manufacturers, third world countries, and makers of portable electronic devices top the list.
The initial American military research focused on creating reliable, lightweight power sources for military and space applications. Because weight and battery life were the main concerns, aluminum became the best choice for the metal “fuel” since it is lightweight, has a very high energy density, and is recyclable. Unfortunately the first aluminum air batteries were very expensive to produce and only found limited application in underwater vehicles and in Special Forces long term emergency packs. Today the military has reinvested in this technology hoping to develop lightweight “personal power stations” that can be carried by ground soldiers as a way of charging and running integrated computer and communication devices while in the field.
Car manufacturers are always looking for ways to reduce the weight of a vehicle without compromising performance or safety. Having a lightweight, high power battery system could potentially reduce the weight of a vehicle up to 500 lbs. and make electricity a viable power source for hybrid engines. Many car companies are looking at Lithium Air batteries, but Toyota has experimented with a Nickel Aluminum air battery that has the potential of reducing the weight of its Prius over 750 lbs.
Some personal aluminum-air fuel cells have the potential to be up to 75 times more energy dense than lithium-ion fuel cells meaning that laptops, cell phones, PDA’s, tablet computers and all other types of personal entertainment devices could use more of their footprints for components and less for battery, reducing the weight and increasing performance.
The most intriguing place that this technology could (and should) be used is in developing regions of the world that do not have ready access to electric grids. Aluminum air battery research has led to the development of large battery banks that could be used to power sanitation and building efforts in third world countries. This can include simple water purification systems , medical diagnostic tools, and even power to whole houses or communities until an infrastructure can be put into place.
How to Make an Aluminum Air Battery
To make your own Aluminum air battery you will need the following easy to obtain materials; aluminum foil, activated charcoal, salt, water, a bowl, paper towel, two clip leads and a DC motor. Once you’ve acquired these materials clear off a place on your kitchen counter begin assembling your battery.
Create a saturated salt solution by filling the bowl with water and then pouring in salt and stirring it. Once you cannot dissolve any more salt in the water, you are done. Spread out a piece of aluminum foil next to the bowl. Fold over a piece of paper towel and soak it in the bowl, then cover the aluminum foil with the towel. Cover the towel with activated charcoal. Place one lead on top of the charcoal and clip the other lead to the aluminum foil. Fold over the aluminum foil to create a package (a burrito if you will) and then attach the leads to the DC motor. Nothing will happen yet. Press down on the aluminum foil and the motor should spin. The harder you press the faster it will spin. When you release the pressure the motor will stop.
Due to the scalable nature of aluminum air batteries they can also be used in small or larger-scale applications. Potential customers could be anyone that needs a failsafe backup electrical system. Grocery stores could use it to keep freezers and coolers running, telecommunication and internet service providers could use it to eliminate the chance of service loss due to brownout situations, and virtually anyone who operates a business that relies on electricity to deliver its product to customers could use an aluminum-air battery as a backup power source.
References
Tesla Aluminum Air Battery (accessed April 2011)
The Aluminum Association:Technology Insights Aluminum Air Fuel Cell Becoming Commercially Viable (accessed April 2011)
The National Academic Press: Specific Power Characteristics of Chemical Batteries (accessed April 2011)
New Energy and Fuel: Battery Research Prospects Grow (accessed April 2011)
Tech Briefs: Create the Future 2011 Design Contest: Alair aluminum-Air Battery (accessed April 2011)
Image Courtesy of Corrosion Doctors