What is chemical energy? What are the sources and uses of chemical energy? Is it renewable after all, or not? Read on to find out.
What is Chemical Energy?
What defines energy is the ability of a system to produce or consume work. Energy exists in many forms but there are mainly two large categories of energy: the potential and the kinetic energy. Forms such as nuclear, solar, wind, chemical and electrical energy belong to either of these categories.
Chemical energy is a form of potential energy that can be stored in the bonds of chemical compounds. As soon as a chemical reaction takes place, the bonds among molecules break and release or absorb this energy, usually in the form of heat. If a reaction releases heat, it is exothermic. If heat is absorbed, the reaction is endothermic.
Sources of Chemical Energy
As mentioned above, chemical energy is stored within the bonds of molecules and when these bonds break, heat is released or absorbed. These molecules are part of chemical compounds or fuels such as coal, oil, biomass, wood, animal waste, natural gas, food, etc. and they all constitute chemical energy sources that produce heat usually through combustion.
Chemical energy sources can be classified into two types, depending on whether they are naturally or artificially prepared and depending on their natural state (solid, liquid, or gaseous):
- Solid Fuel: wood, coal, lignite, etc.
- Liquid Fuel: petrol, diesel, alcohol, kerosene, etc.
- Gas Fuel: natural gas (coal gas, water gas, bio gas)
All these fuels contain hydrocarbons - organic compounds containing carbon and hydrogen - of various molecular weights. These hydrocarbons may be alkanes (methane, ethane, propane, etc.), alkenes (ethene, propene, etc), cycloalkanes, aromatic hydrocarbons and heterocyclic compounds.
Uses of Chemical Energy
Four of the most important uses of chemical energy are described below:
-
The most common use of chemical energy is the food we consume to generate enough energy to live, work, think, etc. Our body is a chemical machine that breaks down the food to produce heat and other forms of energy (electrical for the nerves) in order to sustain all vital functions.
-
Plants convert solar energy into chemical energy through a process called photosynthesis . During this process, carbon dioxide and oxygen are converted into sugar and water with the help of the sun.
Advertisement -
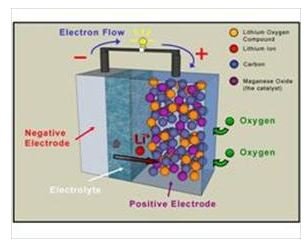
Batteries use chemical energy and convert it to electrical energy to power electric devices, motors, etc. This is achieved through the interaction of specific chemical compounds in order to convert chemical energy to electrical energy. The most common type of battery is the zinc-copper battery. The two materials are used as negative and positive electrode and as soon as the chemical reaction takes places with the aid of the battery’s acid, a flow of electrons is observed. You can find here more information about how these batteries work.
-
 Advertisement
AdvertisementWood, oil, coal and other forms of fossil fuels are burned inside combustion engines to convert chemical energy into heat or motion. These fuels are also burned to produce electrical energy, light and mechanical energy depending on the device and the purpose of use.
Is Chemical Energy Renewable or Not?
The question that arises now is whether chemical energy is renewable or not. Before answering, we should take into account that chemical energy comes from a variety of sources. Some of these energy sources may be renewable such as biomass (plants can grow again) and some of them are not, for example oil, coal etc. Apart from that, some forms of renewable energy such as solar energy, are converted to chemical energy through the photosynthesis process. Taking into account this perspective, it would be rather invalid to classify chemical energy as either renewable or non-renewable energy source.

Sources:
- Chemical Energy by physics.weber.edu
- Chemical Energy Sources by scribd.com
- How is Chemical Energy Used, by tech-faq.com
Image Credits: