Why are Organic Solar Cells so Inefficient?
Organic Solar Cells
On the subject of renewable energy, intensive study of low-cost materials and processes is indispensable in order to come up efficient methods of energy production. Photovoltaic energy conversion is the process of transforming part of the solar electromagnetic spectrum (radiation) into electricity through the use of devices that are called solar cells. The extension of the use of solar cells is dependent on the price of the devices. The use of inorganic materials, such as highly purified silicon, in solar cells cause their production cost to be less competitive with others sources of energy such as coal and natural gas.
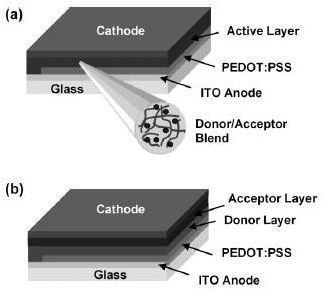
Organic materials used in solar cells can be classified in two main categories: (i) based on conjugated polymers and (ii) based on small organic molecules. The small molecules and polymers used in the organic cells have carbon atoms bonded to each other by alternating single and double bonds. The figure shows typical device structures of the (a) polymer/fullerene solar cell and (b) small-molecule donor-acceptor heterojunction solar cell (PEDOT).
Advantages and Disadvantages of Organic Devices
The process of fabrication of organic solar cells is relatively easy and cheap, since they do not require very high temperatures to be produced, as for the crystalline silicon in inorganic cells. Organic solar cells are made using wet solution processes or dry thermal evaporation of the organic constituents, what makes it possible for them to be deposited on large areas and on different types of substrates, because of the absence of dangling bonds in the structure. They are mechanically flexible.

Organic semiconductors present a high absorption coefficient which is a measure of the amount of electrons that can be excited from the valence to the conduction band. This results from the narrow electronic bandwidth in these organic materials, what causes the effect of absorption to happen in a limited wavelength range of the solar spectrum. Due to the low dielectric constant of organic materials, the electron diffusion lengths are small when compared to inorganic structures.
The introduction of the bulk-heterojunction concept for the photoactive layer resulted in the increase of the energy conversion efficiency. However, in this junction, the absorbed photons do not release the carriers directly, but rather create excitons. Excitons are electron-hole pairs that are not effectively separated from each other. In other to separate the pair it is necessary to have an energy that is higher than the binding energy between electron and hole. The recombination of the excitons that are not split reduces the efficiency of the organic solar cell.
The charge carrier mobility in organic materials is relatively small, and they may be regarded almost as insulators. This situation takes place because they do not have a three-dimensional crystal lattice and also present local disorders and impurities, causing their energy bands to behave differently from the bands of inorganic materials junctions. Other effects that contributes to a small charge carrier mobility are: (i) the weak forces of interaction (van der Waals forces) present in the organic materials that result in small overlap of electronic wavefunctions and (ii) low intrinsic (thermally excited) charge carrier concentration. Regarding the problem of the lattice disorder, the nanoscopic order can be optimized by the preparation conditions.
Other major concerns while producing organic solar cells are the long-term stability and the control of nanomorphology of the organic materials. The stability may be affected by photodegradation of the active materials, but encapsulation techniques can keep the cell from the presence of oxidizing agents.
References
HOPPE, H. et al. Organic Solar Cells: An Overview. J. Mater. Res. 19, 7 (2004).
CNRS. “Renewable Energies: The Promise of Organic Solar Cells.” ScienceDaily, 10 April 2009. Web. 19 November 2010. https://www.sciencedaily.com/releases/2009/04/090409151444.htm
SHROTRIYA, V. et al. Accurate Measurement and Characterization of Organic Solar Cells. Advanced Functional Materials, 16 (2006).
HOPPE, H. Nanoscale Morphology of Conjugated Polymer/Fullerene-Based Bulk-Heterojunction Solar Cells. Advanced Functional Materials 14, 10 (2004).
Further Reading
Bright Hub: Can Multiple Excitons be Produced from One Photon?